The kelvin is a unit of measurement for temperature. It is one of the seven base units in the International System of Units (SI) and is assigned the unit symbol K. The Kelvin scale is an absolute, thermodynamic temperature scale using as its null point absolute zero, the temperature at which all thermal motion ceases in the classical description of thermodynamics.
The reference point that defines the Kelvin scale is the triple point of water at 273.16K (0.01 degrees Celsius). The kelvin is defined as 1/273.16 of the difference between these two reference points.
Absolute zero is the theoretical temperature at which entropy reaches its minimum value. The laws of thermodynamics state that absolute zero cannot be reached using only thermodynamic means.
A system at absolute zero still possesses quantum mechanical zero-point energy, the energy of its ground state. The kinetic energy of the ground state cannot be removed. However, in the classical interpretation it is zero and the thermal energy of matter vanishes.
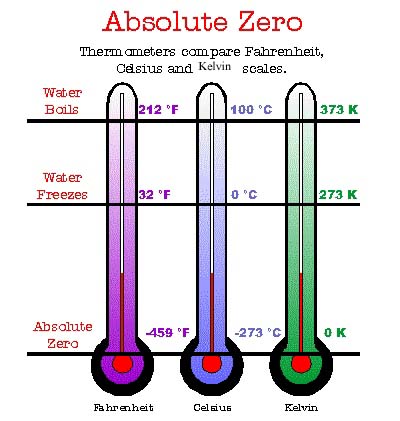
Absolute zero is the null point of any thermodynamic temperature scale. By international agreement, it is defined as 0K on the Kelvin scale and as -273.15°C on the Celsius scale. This equates to -459.67°F on the Fahrenheit scale. Scientists have achieved temperatures very close to absolute zero, where matter exhibits quantum effects such as superconductivity and superfluidity.
Absolute, or thermodynamic, temperature is conventionally measured in kelvins (Celsius-scaled increments) and in the Rankine scale (Fahrenheit-scaled increments) with increasing rarity.
Absolute temperature measurement is uniquely determined by a multiplicative constant which specifies the size of the "degree", so the ratios of two absolute temperatures, T2/T1, are the same in all scales.