CHEM 2414 Basic
Organic and Biochemistry Lec. 4 Lab. 0 Cr. 4
Fall 2005 Section: [21] Time/Day: 4:00-7:50 p.m. T Room: SCI 218
Instructor: Mr. John Taylor
Instructor’s Office: Science 215 Office
Phone: (318) 427- 44357
Cell Phone:
(813) 361-4379 (Monday, Fridays and weekends)
Email:
jtaylor@lsua.edu alternate jtaylor@lsua.info
MAPS Division Office: 473-6591
Text:
General, Organic, and Biochemistry
|
Katherine
J Denniston, TOWSON UNIVERSITY
Joseph J Topping, TOWSON UNIVERSITY
Robert L Caret, SAN JOSE STATE UNIV
|
|
Hardcover, 896 pages
|
|
©2004, ISBN 0072469056
|

Dennison,
Topping, Caret
4th Edition;
Course
Description
(Also offered as BIOL 2414). Prerequisite:
CHEM 1202. A presentation of:
1) the fundamental reaction
capabilities of organic molecules and their functional groups, and
2) the basic principles of
physiological chemistry with an emphasis on their application to problems
encountered in the practice of nursing.
Four hours of
lecture each week.
|
Table of Contents
|
|
General Chemistry (assumed prerequisite)
1 Chemistry: Methods
and Measurement
2 The Composition and
Structure of the Atom
3 Elements, Atoms,
Ions, and the Periodic Table
4 Structure and
Properties of Ionic and Covalent Compounds
5 Calculations and the
Chemical Equation
6 States of Matter:
Gases, Liquids, and Solids
7 Reactions and
Solutions
8 Chemical and
Physical Change: Energy, Rate, and Equilibrium
9 Charge-Transfer
Reactions: Acids and Bases and Oxidation-Reduction
10 The Nucleus,
Radioactivity, and Nuclear Medicine
Organic Chemistry (all chapters covered)
11 An Introduction to
Organic Chemistry: The Saturated Hydrocarbons
12 The Unsaturated
Hydrocarbons: Alkenes, Alkynes, and Aromatics
13 Alcohols, Phenols, Thiols, and Ethers
14 Aldehydes
and Ketones
15 Carboxylic Acids
and Carboxylic Acid Derivatives
16 Amines and Amides
Biochemistry (all chapters covered)
17 Carbohydrates
18 Lipids and Their
Functions in Biochemical Systems
19 Protein Structure
and Function
20 Enzymes
21 Carbohydrate
Metabolism
22 Aerobic Respiration
and Energy Production
23 Fatty Acid
Metabolism
24 Introduction to
Molecular Genetics
Appendixes
A A
Review of Mathematics Applied to Problem Solving in Chemistry
B Table of Formula
Weights
C Determination of
Composition and Formulas of Compounds
D Stereochemistry and Stereoisomers Revisited
E Lipid-Soluble
Vitamins
F Water-Soluble
Vitamins
G Energy Yields from
Aerobic Respiration: Some Alternatives
H Minerals and
Cellular Function
|
Course
Objectives
Students
will:
1. Understand the implications of
structure and functional groups on reactivity, physical behavior and chemical
behavior of organic molecules.
2. Identify
the typical uses of the various classes of molecules.
3. Understand
the outcome of reactions of the various functional groups.
4. Understand
the principles of stereochemistry and their application to reactivity and
biological behavior.
5. Be able to properly name organic molecules and draw
structures from a name.
6. Recognize
biomolecules and reactions typical for them.
7. Understand
the various roles of biological molecules in living systems.
8. Understand
metabolism and regulation of metabolism.
Detailed Topical Outline
0. Review of Chemical Bonding, Molecular
Geometry, Acids & Bases, pH
1. Carbon chains, bonding, and model
building
2. Functional groups: nomenclature,
physical and chemical properties, reactions.
a. Saturated
hydrocarbons
b. Unsaturated
hydrocarbons and aromatics
c. Alcohols,
phenols, thiols and ethers
d. Aldehydes and ketones
e. Carboxylic
acids and derivatives
f. Amines
and amides
3. Biomolecules:
Structure and Function
a. Carbohydrates
b. Lipids
c. Amino
acids and proteins
d. Enzymes
e. Bases,
nucleotides, RNA and DNA
4. Metabolism
a. Carbohydrates
b. Aerobic
respiration and energy
c. Lipid
catabolism
d. Amino
acid catabolism
5. An
introduction to molecular genetics
a. Replication
b. Transcription
c. Translation
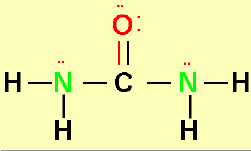
This synthesis of this molecule changed Organic
Chemistry:
Do you know the story?
ATTENDANCE:
Students are expected to
attend class and will be responsible for all material presented. Pretest
quizzes will be administered during every class which is not a scheduled exam
day. These quizzes may not be made up outside of class time.
MAKE-UP POLICY:
Make-up exams are usually not given. In the event of an
unavoidable absence (jury duty, hospitalization, incarceration, and death in
the immediate family), you must contact the instructor, no later than, the day
of the exam in order to discuss what arrangements might be made. A message must
be left on the instructor's e-mail (jtaylor@lsua.edu) if the instructor cannot
be reached. If a makeup is allowed, it must be completed prior to return of the
exam papers completed by the student attending. Missed exams will otherwise
count as 0 points. Student who takes the test on the assigned test day are
guaranteed to receive their graded exam on or before the next exam day after
completion of the new exam, otherwise the student will be assigned a 100% grade
for the un-graded paper.
GRADING:
Exams mainly determine a
student's letter grade. There will be 1000 points possible in the course. The
four hourly exams are worth 100-200 points each. The final exam is worth 200
points. Homework will not exceed 15% of the grade. The approximate grade
distribution is:
900 - 1000 points = A
800 - 899 points = B
700 - 799 points = C
600 - 699 points = D
The instructor reserves the right to make necessary modifications or
adjustments to the point structure above during the semester as necessary,
except the grade level will not change from 90%, 80%, 70% etc. of the total
points.
Exams (Approximate Date):
Exam 1 (T, Sept 13): Chapters. Chapter 11, 12
Exam 2 (T, Oct 4): Chapters 13, 14, 15, 16
Exam 3 (T, Nov 1): Chapters 17, 18, 19, 20
Exam 4 (T, Nov 29): Chapters 21, 22, 23, 24
Final Exam (T, Dec 6, 4:00-7:00 p.m.): All topics-Multiple Choice
Exams will be based on
material covered in the lecture as well as additional reading assignments
outlined on the course calendar.
Daily Quizzes (optional):
Scored pretest quizzes are NOT recorded in the instructor’s
grade book, but must be attached to the Exam the day of the exam to receive the
pretest grade. The scores may be recorded on the attendance sheet. The instructor only records Module Exam
totals and the Final Exam in his grade book. The pretests may
NOT be used during the exam! Samples of each
section (pretest) of each exam may be found on the grading outline on the web
site after the first two weeks of class.
Link to: Chem
2414 Grading Outline
WEB-SITE:
This course uses the
Desire2learn web site giving you access to course information, Email, and
check-your-grade through the Internet. Access the web site at: http://lsua.edu. Your username is your first,
middle and last initial (all in caps) followed by the last 4 digits of your
student ID number. Your password is your student identification number.
OFFICIAL OFFICE HOURS:
(see posting on the instructor’s door for changes and postponements)
Monday: 4:00-6:00 p.m.
Tuesday: 12:30-1:00; 2:15-2:45; 3:30-4:00 p.m.
Wednesday: 3:00-4:00; 5:00-6:00
p.m.
Thursday: 12:30-1:00; 2:15-3:45; 4:30-6:00 p.m.
Friday: 8:30-9:30 a.m.
(Unofficial – anytime I am
in my office)
Instructor’s Right to Change or Modify Grading
Procedures:
This
instructor reserves the right to make changes in this syllabus whenever he
feels it is appropriate to do so. The instructor reserves the right to modify
or change the grading progress as the course proceeds. Any additional course
assignments will substitute for deleted items.
Some may also be modified if not deleted. The instructor will not add major
examinations as a modification and maintain the four exam plus final
requirements
Students with Disabilities:
Qualified students with documented disabilities are
eligible for physical and academic accommodations under the American
Disabilities Act and Section 504 of the Rehabilitation Act of 1973. Students requesting accommodations should
contact this professor during the
first week of class with official documentation of disability
Withdrawal Policy:
Students will be allowed to withdraw from this class
any time during the semester through Tuesay, November 1, 2005 and will receive a grade of
“W”. After this date a letter grade will
be assigned reflecting the student’s performance in the class. Students failing to attend class for two
consecutive weeks are subject to withdrawal by the instructor according to LSUA
policy.
Academic Misconduct:
Academic misconduct or dishonesty such as cheating
and plagiarism is not permitted.
Suspected cases may be reported to the LSUA administration and may
result in failure of an assignment or exclusion from the class. Also, the instructor reserves the right to reassign
work to students if the instructor senses/suspects the work submitted is not
the work of the student. (No questions asked-The instructor may tell the
student to reattempt the work to earn the daily quiz grade or examination grade
or the instructor may assign a zero if second request is made).
Classroom Etiquette:
Students
are expected to conduct themselves as adults in the classroom showing respect
to their classmates. Only persons registered for this class are permitted in
the classroom. As a courtesy to the
instructor and your fellow classmates, cellular
telephones and pagers should be cut off before entering the classroom or
laboratory. Likewise, the instructor sometimes forgets to shut his down
at the beginning of class, so hopefully someone sitting close to the front may
remind the instructor with a hand gesture for him to check his phone,
Studying:
Chemistry
is a cumulative subject. Concepts learned in the first assigned chapter will be
applied in the second, etc. The final exam is cumulative. In order to do well in this course, it is essential to study and work
problems. The following is a list of
study suggestions
1)
Read the text chapters
before the material is covered in class.
2)
Take good notes and review
them daily.
3)
Work all assigned homework
problems at the end of the assigned chapters.
Do not get behind!!!!!!
4)
Work the practice exams that
are available on the web site without looking at the answer key.
5)
Use the interactive web site
for studying.
Procedures
to Evaluate these Objectives
1. In-class and homework problems after concept presentation
2. In class
e-presentation system responses
3. In-class exams with pretests
4. Cumulative final exam

Instructor Requested Information:
During the first week of
class, the student will fill out a 4x6 file card. The instructor has provided a
sample below with his personal data and his block scheduled time. The completion of this card is worth (2 points)
toward the student's final grade
Data Card (4x6 file card): Front Side (Personal Data)
--------------------------------------------------------------------------------------
Name: John Taylor CHEM 1001
Office: Science 215
Address: 6956 England Drive
Alexandria, LA 71303
Telephone: 427-4435 (office) 445-9894 (home till 9/30)
Cell: 813 361-4379 (cell after 9pm Tues-Fri- all day Mon &
weekends)
E-MAIL :
jtaylor@lsua.edu or jtaylor@hccfl.edu
Employment: LSU-Alexandria since 8/15/05
Full time chemistry faculty
Major:
Instructional Technologies Minor:
Chemical Education
Long Term
Goal: Educational Software Developer
Prerequisite:
CHEM 1202 completed
Final grade: A
Software/Computer Literacy: WP: Word
Home Computer: yes Internet ISP: yes or have access
Why are you
taking this course?
--------------------------------------------------------------------------------------








