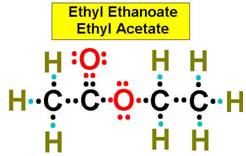
Ethyl Acetate (Ethyl Ethanoate) is a fairly simple organic ester molecule.
It is an Acetic acid molecule (Ethanoic Acid) ( CH3-COOH ) with the acidic hydrogen in the carboxylic acid group ( - COOH )
replaced with an alkyl radical (Ethyl Group) (-CH2CH3).
An Ester results when a carboxylic acid ( R-COOH ) reacts with an alcohol ( R'-OH )
usually in a hydrogen ion catalyst and a water molecule (H-OH) is form, linking the alcohol to the carboxylic acid with an esterfication link ( R-COO-R' )
R and R' stands for any alkyl group.